For cancer patients, sugar-coated cells are deadly
By Anne Ju
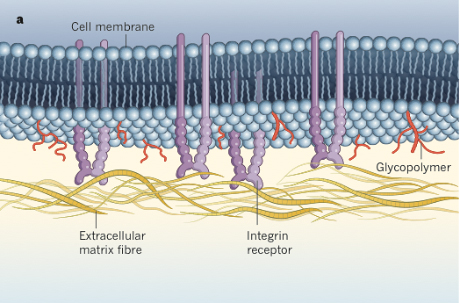
Every living cell’s surface has a protein-embedded membrane that’s covered in polysaccharide chains – a literal sugar coating. On cancer cells, this coating is especially thick and pronounced.
A Cornell researcher has found that a cancer cell’s sugary outside is anything but sweet. The thick, slimy coating that would feel like a slug’s skin is a crucial determinant of the cell’s survival, it turns out. Consisting of long, sugar-decorated molecules called glycoproteins, the coating causes physical changes in the cell membrane that make the cell better able to thrive – leading to a more lethal cancer.
Matthew Paszek, assistant professor of chemical and biomolecular engineering, led the study on glycoprotein-induced cancer cell survival, published online in Nature June 25. Before he joined the Cornell faculty in January, Paszek was a postdoctoral fellow at the Kavli Institute at Cornell for Nanoscale Science. The Nature study was initiated by Paszek and co-author Valerie Weaver at the University of California, San Francisco, and includes other collaborators in bioinformatics, clinical research and chemistry. At Cornell, Paszek’s lab will focus on developing high-resolution microscopy to further study cell membrane-related cancer mechanisms.
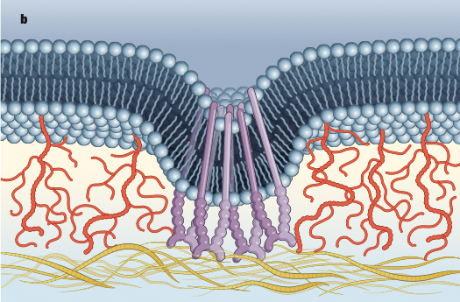
The researchers found that long glycoprotein chains on a cancer cell’s surface cause the cell membrane to push away from its environment and bend inward. This physical change causes adhesion receptors on the cell surface called integrins to clump together. Integrins bind to protein scaffolds in their environment and regulate pretty much everything a cell does – movement, change and growth.
This clustering mechanism causes the integrins to alter the cell’s normal signaling, leading to unchecked growth and survival.
The mechanics of this sugar coating and its subsequent chemical signaling is likely dictated by basic factors like nutrient availability and metabolism – things with implications for diet and genetic makeup, for example, Paszek said. “Changes to the sugar composition on the cell surface could alter physically how receptors are organized,” he said. “That’s really the big thing: coupling the regulation of the sugar coating to these biochemical signaling molecules.”
The paper, “The cancer glycocalyx mechanically primes integrin-mediated growth and survival,” was the subject of a “News and Views” feature in Nature. Paszek’s work was supported by the Kavli Institute at Cornell for Nanoscale Science.
Media Contact
Get Cornell news delivered right to your inbox.
Subscribe